Servicios Personalizados
Articulo
Latin American applied research
versión On-line ISSN 1851-8796
Lat. Am. appl. res. vol.45 no.1 Bahía Blanca ene. 2015
Comparison of Cr(VI) adsorption onto polymer-based magnetic hybrid adsorbents by nonlinear isotherm and kinetic models
E. Bilgin simsek†, I.G .Buyruklardan Kaya‡ and U. Beker‡
† Chemical and Process Eng. Department, Yalova University, Yalova, 77200, Turkey. esrabilgin622@gmail.com
‡ Chemical Eng. Department, Yildiz Technical University, Istanbul, 34210, Turkey ubeker@gmail.com
Abstract— In the present study, polyglycidyl methacrylate graft copolymer (PG) and ethylene glycol dimethacrylate based polymers (EG) were synthesized and modified by iron oxides in order to have magnetic form (PG-M and EG-M). Batch hexavalent chromium (Cr(VI)) adsorption experiments were carried out as a function of pH. Adsorption data were applied to Langmuir, Freundlich, Dubinin-Raduskevich, Sips, Toth and Redlich-Peterson isotherm models by utilizing non-linear techniques. The Dubinin-Radushkevich isotherm model was found to be the most suitable one for PG-M sample, while Langmuir model fitted best to the experimental data of EG-M sample. The contact time needed for both adsorbents was relatively short, ranging from 1 to 30 min. Approximately 73% of Cr(VI) was rapidly removed by PG-M within ten minutes; and for EG-M sample, 88% of Cr(VI) was removed at the end of five minutes. The rate of the adsorption is governed by a pseudo-second order rate equation.
Keywords— Polymer; Magnetic Separation; Cr(VI) Adsorption; Nonlinear Isotherm; Kinetic Model
I. INTRODUCTION
Chromium (Cr) is considered as a high priority environmental pollutant and it is on the top-priority list of toxic pollutants defined by the U.S. Environmental
Protection Agency (EPA). According to the EPA
reports, 29 million pounds of chromium is discharged from rainwater, and almost 100,000 pounds of chromium is released from surface water discharges per year (Luther et al., 2013). Large amount of wastewater containing chromium has been released into the environment due to the erosion of certain rocks and volcanic eruptions and due to the industry processes such as electroplating, wood protection, leather tanning, magnetic tapes, pigments, metal finishing, electrical and electronic equipment, textile industries and chromate preparation (Singh et al., 2011; Kimbrough et al., 1999). Chromium essentially exits in the environment in two forms namely hexavalent chromium [Cr(VI)] and trivalent chromium [Cr(III)]. Especially Cr(VI) is considered as one of the top 16 toxic pollutants because of its carcinogenic and teratogenic properties and threatens the natural life and public health (Bansal et al., 2009). Exposure to Cr(VI) causes health problems of epigastria, nausea, vomiting, severe diarrhea, dermatitis, kidney, liver and gastric damage (Yao-Xing et al., 2011). The permitted maximum level of Cr(VI) into the inland surface water and potable water are 0.1 mg/L and 0.05 mg/L respectively. Therefore, Cr(VI) must be removed from contaminated-water before discharging into the environment in order to protect the living organisms (Nethaji et al., 2013).
Several kinds of material were used as adsorbents for Cr(VI) removal, such as activated carbon, polymers, sands, zeolites, agriculture waste, etc. Recently functionalized polymers have been widely used as effective adsorbents in remediation of water according to their mechanical properties and capability of numerous functionalization possibilities. However, these kinds of adsorbents suffer from a common problem that it needs a next separation process from the solution, which increases the operation cost. Therefore, studies have focused on preparation new magnetic hybrid adsorbents using iron oxides.
Iron (oxy-hydr) oxides have been widely used adsorbents for chromium removal due to their excellent removal capacity. Compared with traditional adsorbents, they have extremely high surface area to volume ratio and short diffusion rate, which resulted in high removal efficiency and favorable kinetics for selective sorption. However, these tiny particles cannot be used in fixed-bed columns or any plug-flow-type configuration due to excessive pressure drops. Supporting of high-capacity magnetite nanoparticles into/onto porous matrix of larger size such as functionalized polymeric materials was proven to be effective method in wastewater treatment. The polymer-based magnetic hybrid adsorbents are also good candidates for selective adsorption properties with ease of phase separation, less susceptibility to fouling by natural organic matters, and durability (Bilgin Simsek et al., 2012).
The objective of present study is to investigate the Cr(VI) adsorption efficiency of two polymer-based magnetic hybrid adsorbents. For this reason, polyglycidyl methacrylate graft copolymer (PG) and ethylene glycol dimethacrylate based polymers (EG) were magnetically activated by iron oxides. Batch adsorption experiments were carried out as a function of pH. Adsorption data were applied to the Langmuir, Freundlich, Dubinin-Raduskevich, Sips, Toth and Redlich-Peterson isotherm models. The Cr(VI) adsorption kinetics were tested by using pseudo-first-order, pseudo-second-order, intra-particle diffusion and Elovich kinetic models.
II. METHODS
A. Synthesis of Polymer-Based Magnetic Hybrid Adsorbents
Polyglycidyl methacrylate graft copolymer (PG) was synthesized by using atom transfer radical polymerization technique (Senkal et al., 2007). Ethylene glycol dimethacrylate copolymer beads were produced by the suspension polymerization method (Senkal et al., 2006).
The magnetic hybrid sorbent were prepared by co-precipitating Fe2+ and Fe3+ ions in NaOH solution by treating under hydrothermal conditions. The initial molar ratio of Fe3+:Fe2+ = 2:1 was chosen for the production of Fe3O4. Defined amount of polymer beads were added to aqueous solution containing FeCl2 and FeCl3 under vigorous stirring at 338 K. NaOH was added drop-wise into the solution till pH 10-11. Then, the precipitate was separated by an external magnetic field and rinsed with distilled water until neutral pH. The resultant magnetic adsorbents prepared by using PG and EG polymer beads were denoted as PG-M and EG-M, respectively.
B. Characterization
Potentiometric titration data for the adsorbents were obtained using a method described by Helfferich (1995). Basic titration data was transformed into proton-binding isotherms using a proton balance equation and a theoretical blank reference. The electrophoretic mobilities of the equilibrated samples were measured using a Malvern Instruments Zetasizer 3000HSA. XRD measurement was performed on a Philips Panalytical XPert Pro X-ray diffractometer using CuKα radiation. Magnetic characterization of PG-M and EG-M sorbents was conducted with magnetometer (Micromag 2900, Measurements, Princeton, USA).
C. Adsorption Experiments
Adsorption experiments were carried out using batch equilibration techniques. The effect of solution pH (2 .0, 4.0, 6.0, 10.0) on adsorption was investigated with initial concentration of 30 mg/L at 298 K. Adsorption isotherms were obtained by varying amount of adsorbents. The suspensions were shaken at 130 rpm in a platform shaker at ambient temperature. pHs of the suspensions were kept constant during adsorption experiments and adjusted with NaOH or HCl twice a day. Once the equilibrium was established, suspension was filtered with a blue band filter paper. Experimental adsorption capacities were calculated by using the following equation:
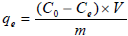 | (1) |
where qe is adsorption capacity (mg/g), C0 and Ce are the Cr(VI) initial and equilibrium concentrations (mg/L), respectively. V is the volume of the Cr(VI) solutions and m is the weight of adsorbent (g).
Kinetic experiments were conducted in a 500 mL glass vessel with of initial Cr(VI) concentration of 30 mg/L. Equilibrium Cr(VI) concentration in the aqueous solution was determined by UVvis spectrophotometer (Analytic Jena Specord 40 UV) at 540 nm.
The relationship between adsorbed Cr(VI) and the concentrations at equilibrium was described by two- and three-parameter isotherm models: Freundlich, Langmuir, Dubinin-Radushkevich, Redlich-Peterson, Sips, and Toth using the Statistica 8.0 programme. The kinetic data were applied to pseudo-first-order, pseudo-second-order, intra-particle diffusion and Elovich models.
III. RESULTS AND DISCUSSION
A. Characterization
The proton binding curve which crosses the pH axis is called the point of zero charge (PZC) and defined as the pH value at which the net surface charge of the adsorbent is zero and changes depending on the type and number of functional groups present on the adsorbent (Bilgin Simsek et al., 2012). The point of zero charge (pHPZC) of PG-M and EG-M were about 6.3 and 6.8, respectively (Table 1). Below the pHPZC, the adsorbent surface has positive charge and affinity to anions. On the other hand, above the pHPZC adsorbent surface is negatively charged, and cation adsorption occurs. Dissociation reactions of magnetic polymers surface can be shown by Equations (2-4):
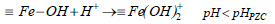 | (2) |
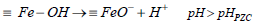 | (3) |
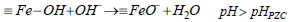 | (4) |
An important tool used to characterize the electrokinetic behavior of a solid-liquid interface is the electrophoretic mobility. The pH value at this point is called the isoelectric point (IEP) of the interface. The IEP for PG-M sample is 8.5, while that for EG-M is 7.8. The reason for higher pHIEP values of samples is probably due to the fact that their surfaces have much positive groups of iron oxides.
According to the magnetic characterization (Fig.1); the saturation magnetization value of EG-M was found as 40.7 emu/g, while that of for PG-M was obtained as 21.6 emu/g. The magnetization values were found higher for both samples indicating ferromagnetic form has covered the polymeric surfaces.
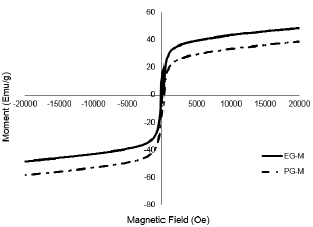
Fig. 1. Magnetization curves of samples
The XRD pattern of both samples (data not shown) indicated characteristic peaks at about 2θ=30.16o, 35.63o, 42.24o and 62.58o related to the Fe3O4 phases. The patterns also showed that the surface of magnetically activated sorbent was coated pure Fe3O4.
B. Adsorption Experiments
It is well known that pH is a critical and an important parameter in adsorption process as it influences chemical speciation of metal ions and active functional sites on surface of sorbent (Kragovic et al., 2012; Chutia et al., 2009). The maximum Cr(VI) adsorption capacities of PG-M and EG-M were determined at pH 4 as 106.2 and 78.2 mg/g, respectively (Fig. 2). The magnetic hybrid adsorbents have considerably high Cr(VI) sorption capacities when compared with other capacities of similar adsorbents reported in the literature (Hu et al., 2005; Bayramoglu and Arica, 2008; Yuan et al., 2009; Gang et al., 2010).
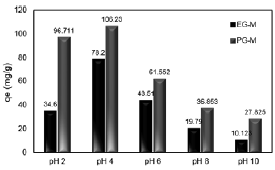
Fig. 2. Effect of pH on Cr(VI) adsorption
Cr(VI) ions exist in several stable forms in aqueous solution. CrO42− is predominant in basic solutions, while HCrO4− and Cr2O72− are predominant at pH 2.0-6.0. It is possible that, under acidic conditions amine groups in PG-M and EG-M samples might be protonated and the HCrO4− anions attract -NH3+ groups. Therefore, electrostatic attraction could be responsible for that high adsorption capacity at this pH range. Moreover, due to the combined effects of Fe oxides and amine groups, -under acidic pH- higher Cr(VI) adsorption occurred via electrostatic interaction and complex formation with Lewis acid base reaction. Fe3O4 has FeOH2+ surface groups in aqueous solution and complex formation might be occurred between FeOH2+ and HCrO4−/CrO42−. FeOH2+ surface groups act as electron acceptors (Lewis acid) while chromium species act as electron donors (Lewis base).
With increasing pH, maximum adsorption capacity of PG-M reduced to 27.82 mg/g at pH 10, while that of EG-M was found as 10.12 mg/g. The decrease in removal capacity above pHPZC is attributed to the electrostatic repulsion between negatively charged surface groups and chromium ions and to the competition with OH− ions in the reaction mixture. Moreover, the amine groups are dissociated at about pH of 10; hence, electrostatic repulsion is occurred resulting in declining adsorption capacity. It can be also mentioned that, increasing pH leads to shift the formation of HCrO4− to CrO42−, which needed two active sites because of two minus charges of it. So, the interaction between the adsorbent surface and the CrO42− anions are weaker.
Moreover, the effect of three different temperature 298, 308 and 318 K on Cr(VI) adsorption was investigated. The adsorption affinity of samples was increased by increasing temperature indicating exothermic nature of process. Enthalpy change of Cr(VI) adsorption was calculated as −24.65 kJ/mol and −11.84 kJ/mol for EG-M and PG-M adsorbents, respectively. These low values of enthalpy change demonstrated that the electrostatic interaction is the main mechanism for Cr(VI) adsorption onto both sorbents.
C. Isotherm Models
In order to evaluate the conformity of applied isotherms to experimental data, the coefficient of determination (R2) and Chi-square values (χ2) were calculated (Table 1) and experimental and theoretical capacities were given in Fig 3 and 4.
Table 1. Isotherm constants for Cr(VI) adsorption onto adsorbents (pH 4, C0: 30 mg/L)
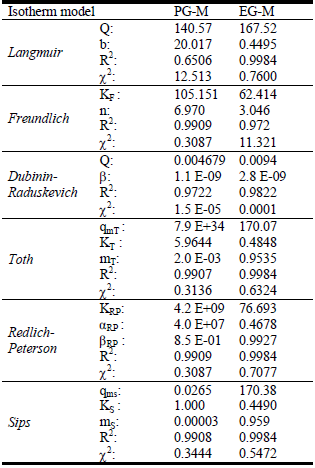
Fig. 3. Adsorption isotherms of EG-M (---: Experimental,  : Langmuir,
: Langmuir,  : Sips,
: Sips, 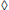 : Toth, ×: Freundlich, ····: R-P, Δ: D-R)
: Toth, ×: Freundlich, ····: R-P, Δ: D-R)
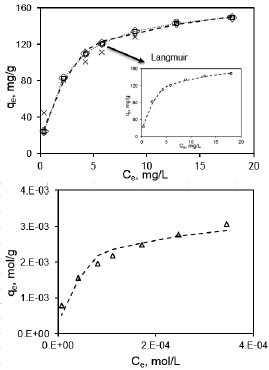
Fig. 4. Adsorption isotherms of EG-M (---: Experimental,  : Langmuir,
: Langmuir,  : Sips,
: Sips,  : Toth, ×: Freundlich, ····: R-P, Δ: D-R).
: Toth, ×: Freundlich, ····: R-P, Δ: D-R).
The obtained results for PG-M showed that the best-fitted adsorption isotherm models were in the order of: Dubinin-Raduskevich > Sips > Redlich-Peterson > Toth > Freundlich > Langmuir, on the basis of high regression coefficients (R2) and lower χ2 values. Among the two-parameter models Dubinin-Raduskevich -which assumes constant sorption potential- and among the three-parameter models Sips isotherms describe with satisfaction of the equilibrium data.
However, for EG-M sample Langmuir model fitted best to the experimental data indicating homogenous reactions occur during adsorption process.
D. Adsorption Kinetics
The controlling mechanism of the adsorption process was investigated by fitting the experimental data with pseudo-first-order (Fig. 5), pseudo-second-order (Fig. 6), intra-particle diffusion (Fig. 7) and Elovich (Fig. 8) kinetic models. According to experimental and theoretical kinetic data in Table 2, the experimental results obtained for the Cr(VI) adsorption onto PG-M and EG-M sorbents were found to obey the second-order kinetic and the intra particle diffusion. The theoretical capacity values estimated from the first-order kinetic model gave significantly different values compared to experimental values (qexp), and the correlation coefficients were also found to be lower. This indicates that pseudo first-order equation might not be sufficient to describe the mechanism of Cr(VI) adsorption system.
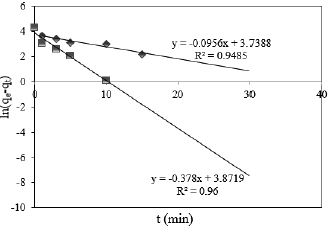
Fig. 5. Pseudo first order kinetic model for Cr(VI) adsorption onto PG-M ( ) and EG-M (
) and EG-M ( )
)
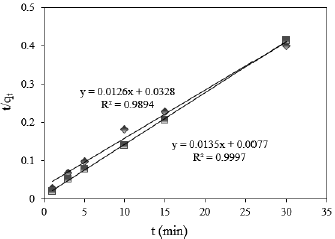
Fig. 6. Pseudo second order model for Cr(VI) adsorption onto PG-M ( ) and EG-M (
) and EG-M ( )
)

Fig. 7. Intra-particle diffusion model for Cr(VI) adsorption onto PG-M ( ) and EG-M (
) and EG-M ( )
)
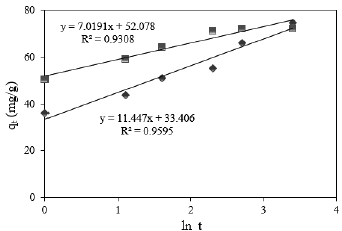
Fig. 8. Elovich model for Cr(VI) adsorption onto PG-M ( ) and EG-M (
) and EG-M ( )
)
Table 2. Kinetic model parameters of samples
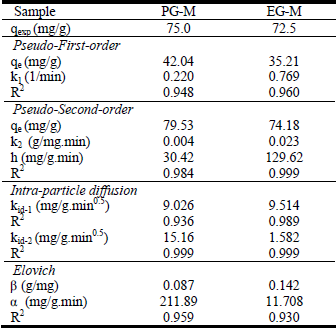
The correlation coefficients (R2) for the pseudo second-order kinetic model are in the range of 0.994-0.999 and the theoretical qe (cal) values were closer to the ex perimental qe (exp) values. The pseudo-second-order model is based on the assumption that the rate-determining step might be a chemical sorption involving valence forces through sharing or exchange of electrons between adsorbent and adsorbate.
Moreover, the plots of qt versus t0.5 indicates that the intra-particle diffusion models well the Cr(VI) adsorption processes (Fig. 7). The correlation coefficients of intra-particle model were in the range of 0.994-0.999 for both rapid and slow reactions. It is clear from the figure that two linear consecutive steps are observed: first linear portion (fast reaction) and second linear part (slow reaction). In the first portion, approximately 73% of Cr(VI) was rapidly uptaken by PG-M within ten minutes; and for EG-M sample, 88% of Cr(VI) was removed within five minutes. This could be attributed to the immediate utilization of the most readily available adsorbing sites on the adsorbent surface (Kumar et al., 2009). The second phase indicates very slow diffusion of the adsorbate from the surface site into the inner pores.
The Elovich equation plots for the Cr(VI) adsorption are shown in Fig. 8. The plots are fairly linear. The R2 values are quite high and show good fit according to the R2 value of 0.93 and 0.95 for EG-M and PG-M, respectively. Thus, it could be said that Elovich equation is a good model for the sorption process.
IV. CONCLUSIONS
In the present study, polymer-based magnetic hybrid sorbents as PG-M and EG-M have been successfully synthesized using co-precipitation method. The magnetically activated adsorbents showed good adsorption capacity for Cr(VI) ions. The maximum adsorption capacity was observed under acidic pH. It was concluded that the electrostatic attraction could be responsible for that high adsorption capacity at this pH range. The equilibrium distributions of Cr(VI) ions between magnetic hybrid sorbents and liquid phases were modelled by isotherm equations. The Dubinin-Radushkevich isotherm model was found to be the most suitable one for PG-M sample, while Langmuir model fitted best to the experimental data of EG-M sample. The contact time needed for both adsorbents was relatively short, ranging from 1 to 30 min. The rate of the adsorption is governed by a pseudo-second order rate equation indicating that the rate-determining step might be chemical sorption.
Considering with the conventional separation techniques, magnetic separation is an advantageous method due to its rapidness, efficiency and simplicity.
REFERENCES
1. Bansal, M., D. Singh and V.K. Garg, "A comparative study for the removal of hexavalent chromium from aqueous solution by agriculture wastes' carbons," J. Hazard. Mater., 171, 83-92 (2009).
2. Bayramoglu, G. and M.Y. Arica, "Adsorption of Cr(VI) onto PEI immobilized acrylate-based magnetic beads: isotherms, kinetics and thermodynamics study," Chem. Eng. J, 139, 20-28 (2008).
3. Bilgin Simsek, E., D. Duranoglu and U. Beker, "Heavy Metal Adsorption by Magnetic Hybrid-Sorbent: An Experimental and Theoretical Approach," Sep. Sci. Tech., 47, 1334-1340 (2012).
4. Chutia, P., S. Kato, T. Kojima and S. Satokawa, "Arsenic adsorption from aqueous solution on synthetic zeolites," J. Hazard. Mater., 162, 440-447 (2009).
5. Gang, Z.Y., S.H. Yu, P.S. Dong, H.M. Qina, "Synthesis, characterization and properties of ethylenediamine-functionalized Fe3O4 magnetic polymers for removal of Cr(VI) in wastewater," J. Hazard. Mater.,182, 295-302 (2010).
6. Helfferich, F., Ion Exchange, Dover Publications, (1995).
7. Hu, J., G. Chen, I. M. C. Lo, "Removal and recovery of Cr(VI) from wastewater by maghemite nanoparticles," Water Res., 39, 4528-4536 (2005).
8. Kimbrough, D.E., Y. Cohen, A.M. Winer, L. Creelman and C. Mabuni, "A Critical Assessment of Chromium in the Environment," Crit. Rev. Environ. Sci. Technol., 29, 1-46 (1999).
9. Kragovic, M., A. Dakovic, Z. Sekulic, M. Trgo, M. Ugrina, J. Peric, G.D. Gatta, "Removal of lead from aqueous solutions by using the natural and Fe(III)-modified zeolite," App. Surf. Sci., 258, 3667-3673 (2012).
10. Kumar, E., A. Bhatnagar, M. Ji, W. Jung, S.H. Lee, S.J. Kim, G. Lee, H. Song and J.Y. Choi, J.S. Yang, and B.H. Jeon, "Defluoridation from aqueous solutions by granular ferric hydroxide (GFH)," Water Res., 43, 490-498 (2009).
11. Luther, S., N. Brogfeld, J. Kim and J.G. Parsons, "Study of the thermodynamics of chromium(III)and chromium(VI)binding to iron(II/III)oxide or magnetite or ferrite and magnanese(II) iron(III)oxide or jacobsite or manganese ferrite nanoparticles," J. Colloid Interface Sci., 400, 97-103 (2013).
12. Nethaji, S., A. Sivasamy and A.B. Mandal, "Preparation and characterization of corn cob activated carbon coated with nano-sized magnetite particles for the removal of Cr(VI)," Bioresour. Technol., 134, 94-100 (2013).
13. Senkal, B.F. and E. Yavuz, "Crosslinked poly(glycidyl methacrylate)-based resin for removal of mercury from aqueous solutions," J. Appl. Polym. Sci., 101, 348-352 (2006).
14. Senkal, B.F., F. Bildik, E. Yavuz and A. Sarac, "Preparation of poly(glycidyl methacrylate) grafted sulfonamide based polystyrene resin with tertiary amine for the removal of dye from water," React. Funct. Polym., 67, 1471-1477 (2007).
15. Singh, K.P., A.K. Singh, S. Gupta and S. Sinha, "Optimization of Cr(VI) reduction by zero-valent bimetallic nanoparticles using the response surface modeling approach," Desalination, 270, 275-284 (2011).
16. Yao-Xing, L., Y. Dong-Xing, Y. Jun-Mei, L. Quan-Long and O. Tong, "Electrochemical removal of chromium from aqueous solutions using electrodes of stainless steel nets coated with single wall carbon nanotubes," J. Hazard. Mater., 186, 473-480 (2011).
17. Yuan, P., M. Fan, D. Yang, H. He, D. Liu, A. Yuan, J. Zhu, T. Chen, "Montmorillonite-supported magnetite nanoparticles for the removal of hexavalent chromium [Cr(VI)] from aqueous solutions," J. Hazard. Mater., 166, 821-829 (2009).
Received: January 7, 2014.
Accepted: August 7, 2014.
Recommended by Subject Editor: Marcelo Seckler.